Investigators at Hackensack Meridian John Theurer Cancer Center Are Leading Transformative Work in T-Cell Therapy for Cancer Patients
Renowned lymphoma experts at Hackensack Meridian John Theurer Cancer Center at Hackensack University Medical Center are leading a full range of trials at various stages of development – from studies granted accelerated approval by the U.S. Food and Drug Administration (FDA) to long-term follow-ups and from multiple myeloma to leukemia, including:
- The ZUMA 2 trial – The FDA granted accelerated approval for brexucabtagene autoleucel (TECARTUS™, formerly KTE-X19) as the first and only CAR T-Cell therapy for patients with mantle cell lymphoma (MCL) relapsed or refractory to prior treatments. Investigators from John Theurer Cancer Center participated in the pivotal trial on which the approval was based, which assessed the safety and effectiveness of brexucabtagene autoleucel in patients with relapsed or refractory MCL who had received up to five prior regimens of treatment and had exhausted all other therapies. That transformative study showed that 93 percent of patients responded to treatment, with 67 percent achieving a complete response (no evidence of disease).
- Ibrutinib plus lenalidomide and rituximab has promising activity in relapsed and refractory non-germinal center B-cell like DLBCL.
- Lifileucel (LN-144) or tumor infiltrating lymphocytes (TIL) therapy – an investigational immunotherapy.
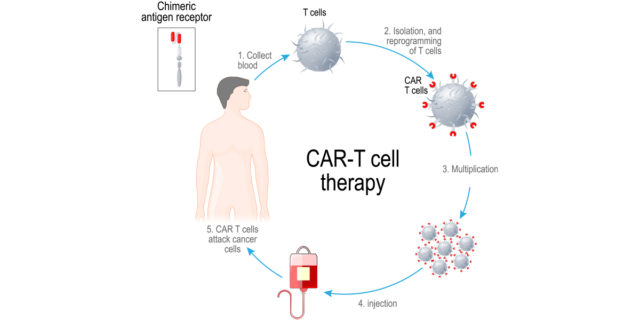
- Chimeric Antigen Receptor T-cell (CAR-T) Therapy – directed against B-cell maturation antigen (BCMA) in participants with multiple myeloma (CARTITUDE-2).
- CYNK-001 – the only cryopreserved, allogeneic, off-the-shelf, natural killer (NK) cell therapy being developed from placental hematopoietic stem cells as a potential treatment option for various hematologic cancers, solid tumors, and infectious diseases.
- Placental-derived Natural Killer (PNK) Cells.
Along with these groundbreaking clinical trials, John Theurer Cancer Center continues to develop and execute crucial cancer research, with 13 trials currently in progress.
