Hackensack University Medical Center Physicians Usher Breakthrough Robotic Technology Into Orthopedic Practice
Orthopedic surgeons pioneer robotic knee surgery techniques ushered into practice
Robotic knee surgery is safer and more precise thanks to the creative contributions of Hackensack Meridian Health physicians.
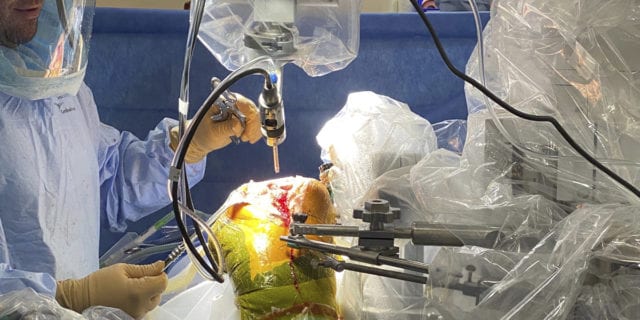
Harlan Levine, M.D., and Gregg Klein, M.D., of Rothman Orthopaedics, were part of an elite global team that developed the ROSA Robotic surgery for total knee replacement. The Zimmer Biomet ROSA Robotic System uses X-rays, instead of a CT-scan, to create a 3D model of the patient’s knee for a personalized implant. This approach also limits the radiation exposure for patients during their scans, while still offering the benefits of robotic surgery, including accurate placement and alignment of implants and less pain for patients.
Hackensack University Medical Center was the first hospital in New Jersey and the first in the northeastern United States to perform total knee replacement surgery using the Zimmer Biomet ROSA system in 2019. Today the center is a center of excellence for training other surgeons in ROSA robotic approach.
In February 2020, Hackensack University Medical Center achieved another robotic surgery milestone: the first FDA approved total knee replacement using the THINK Surgical Inc. TSolution One® Total Knee Application performed by Yair D. Kissin, M.D. The system, which is manufactured by Fremont, California-based THINK Surgical, Inc., features combined 3D surgical planning with a computer-assisted robot to more precisely prepare and implant a patient’s joint.
“Surgeons still have to place the actual implant, but being able to put it in a more correct position can lead to better outcomes for patients and less pain because there’s less soft-tissue manipulation during the surgery,” says Dr. Kissin, adding that most of the 1,500 patients who undergo total knee replacement at the hospital each year would be good candidates for the robotic procedure.
Hackensack University Medical Center was the first facility in the United States to acquire and offer patients undergoing total knee replacement the TSolution One® Total Knee Application for use in total knee arthroplasty (TKA) since the system received clearance from the U.S. Food and Drug Administration in October 2019. Five surgeons, including Dr. Kissin, at four centers nationwide participated in the investigational clinical trial, which confirmed the safety and efficacy of the TSolution One® Total Knee Application.
